Continuing a legacy: Vitamin D research in the 21st century
The legacy of vitamin D research in the Department of Biochemistry at the University of Wisconsin–Madison runs deep — almost as deep as the vitamin’s involvement in a multitude of important processes in the body, such as bone health, muscle strength, the absorption of other vitamins and minerals, and more.
Renowned vitamin D researcher and biochemistry professor Harry Steenbock would have been 130 this year — he was born Aug. 16, 1886 in Charlestown, Wis. — and in his honor the Department of Biochemistry talked to a couple of its researchers about how far vitamin D research has come since his death in 1967. Vitamin D research started here with biochemistry faculty like Steenbock and Elmer McCollum, but it certainly didn’t end when they left.

In these more modern times, a few current faculty members are prominent in vitamin D research in the Department of Biochemistry, which is part of the College of Agricultural and Life Sciences. Professor emeritus Hector DeLuca, one of Steenbock’s last students, continues his work on the vitamin D pathway to this day. Another example is professor Wes Pike, who has taken his work with the vitamin down other avenues, ones that focus at the molecular level to determine mechanisms through which the vitamin regulates the expression of genes — and in doing so controls the numerous biological activities that have been attributed to the vitamin.
Many stories have been told about Steenbock’s work on vitamin D. He and Charles Slichter, the dean of the Graduate School at the time, conceived the Wisconsin Alumni Research Foundation (WARF) in 1925, pioneering the idea that money earned through patents should go back into research at the university. His work also led to a method for adding vitamin D to foods, curing the disease rickets worldwide. (For a refresher, or if you’re new to the exciting field of vitamin D, see here and here.) But, how far has vitamin D come since Steenbock’s time? How have new technologies aided UW–Madison scientists in their research on vitamin D?
“Vitamin D’s story is far from over,” DeLuca says. “Since I took over Steenbock’s lab we have described the pathway the vitamin takes in the body. Researchers like Wes have begun to take a look at gene expression and genomic modifications, really getting to the molecular level.”

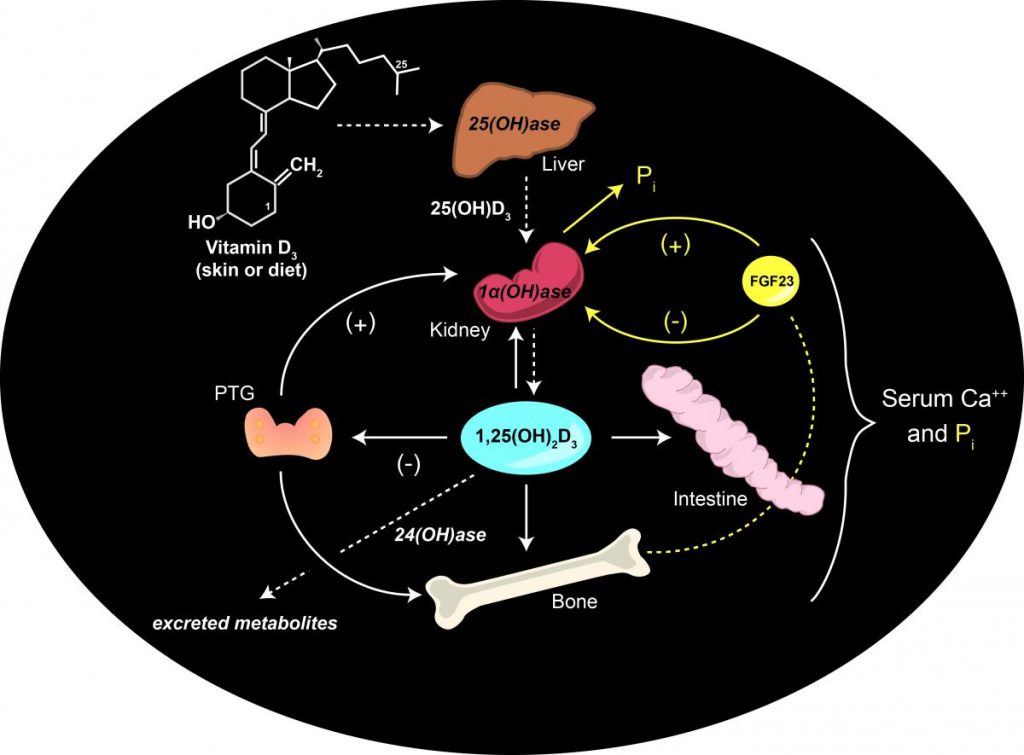
DeLuca continued Steenbock’s line of work after he retired in the ‘50s and sought the “how” and “why” behind vitamin D curing rickets. He started by investigating what happens to vitamin D when it enters the body. The first thing he and his team noticed was that it seemed vitamin D disappears when it enters the body. They quickly discovered that vitamin D obtained via the sun or diet is inactive and must be metabolized by the liver and kidneys into different forms before it can be utilized by the body.
“We developed a research program to isolate and identify these active forms of vitamin D in the mid ‘60s,” DeLuca recalls. “We would isolate enough of the active form and give it back to animals. Because it was potent and acted very fast we knew we were getting somewhere.”
It eventually became known, through DeLuca’s work and others’, that vitamin D goes through two conversions before it is utilized by the body. The first became known as calcidiol (25-hydroxyvitamin D3), and the next form, which is utilized by the body, is calcitriol (1,25-dihydroxyvitamin D3). The numbers designate their particular chemical structure. DeLuca was the first scientist to isolate and fully identify both 25 hydroxyvitamin D3 and 1,25 dihydroxyvitamin D3 forms and patented the structures and methods of synthesis, as well as application to disease.
“That is arguably the biggest major discovery my lab has ever made,” says DeLuca, who has more than 1,500 patents with WARF, which have generated an excess of $500 million in royalties over the past 30 years. “So then we filled in and found out how it’s regulated and we described the vitamin’s path through the endocrine system, and together with other groups discovered the receptor for vitamin D so we could really understand how it works at the physiological level.”
To try to battle diseases caused by vitamin D deficiencies, DeLuca worked with clinical investigators on therapeutics that he patented through WARF. His story with vitamin D continued as he worked to discover why some individuals still develop a vitamin D deficiency and found their bodies lack the ability to convert one form of the vitamin to the other or lack the vitamin D receptor. Three examples of top drugs on the market that sprang from DeLuca’s research are Calcijex, Zemplar and Hectoral, which replace the vitamin D hormone a patient’s body cannot make itself.
Beth Werner, who works with intellectual property at WARF, says DeLuca continues to disclose inventions and further research on the vitamin.
“Vitamin D has a rich history on campus and Hector has a large number of vitamin D analogs that could be tested in many different kinds of research,” she says. “UW-Madison has been a world leader in vitamin research historically. Hector has ensured that the university has remained one of the world’s leaders in vitamin D for the last five or six decades.”
In more recent years, DeLuca has continued to unlock the secrets of the vitamin D pathway and how it can be used as a therapeutic. In 2001, Pike joined the Department of Biochemistry, continuing the department’s legacy of vitamin D research.
Pike brings new and different research and laboratory techniques aimed at understanding the actions of vitamin D and how the molecule interacts at the molecular level to influence the output of specific genes, ultimately impacting which proteins are made. As a graduate student in Arizona, Pike participated in the identification of the vitamin’s cellular receptor and characterized its regulatory properties and with others ultimately cloning the gene. After graduate school, he spent time at Baylor University, a pharmaceutical company, and a university in Ohio, all before joining UW–Madison.

Pike’s goal is to uncover where vitamin D and its receptor binds to DNA on the genome, identify target genes, and then to determine how this binding activity results in altered gene expression. At first glance, it seems logical that the binding locations would be very close to the genes it affects, and so scientists used laboratory techniques that enabled them to focus on regions on the genome that were close to these genes.
However, using two cutting-edge techniques called chromatin immunoprecipitation and parallel DNA sequencing, the Pike Lab is now able to get a less biased glimpse at the locations of these regulatory binding sites and thus determine more precisely the genes that are affected. In essence, instead of guessing which genes may be regulated by vitamin D, he and his team are able to effectively analyze the entire genome of vitamin D sensitive cells. What they’ve found has turned the tables on vitamin D and gene regulation research.
“The advances over the last 15 years have been striking,” Pike says. “We have found new binding sites and new genes but also shown how some of the past research in this area has been incomplete due largely to the methods that were used.”
Recently, Pike has started working almost exclusively “in vivo,” meaning in living organisms. In Pike’s case, the model organism has been the mouse. He says this model and the methods utilized have allowed his team to obtain results that better reflect reality when compared to experiments on cells cultured on plates in the lab. Using a new technology called CRISPR/Cas9, his team can quickly edit the genome of mice very precisely so that the regulation of genes of interest to the lab can be studied.
“The gist is that we have been able to apply these new techniques to the study of vitamin D action very early on, and as a result have been able to forge ahead in a very meaningful way,” he explains. “This approach has completely revolutionized the way we study gene regulation, and has allowed us to discover entirely new principles. It’s all working out fantastically.”
Using these techniques, Pike’s lab has recently revealed that while stem cells in bone marrow are genetically predisposed to be bone cells, they are still capable of becoming fat cells and even trans-differentiating back into bone cells via what is called epigenetic plasticity. They have also recently investigated the role of vitamin D in the calcification, or hardening, of plagues in atherosclerosis.
While Pike and DeLuca’s research is similar, there are some key differences that allow them to continue two independent yet overlapping lines of research in the department. Pike is focusing on mechanisms of vitamin D action, while DeLuca continues to advance on paths of continued discovery that he embarked upon in the ‘50s.
“Wes almost works entirely with 1,25 D3,” DeLuca explains. “He’s working at that last step. What is this doing to a tissue or a gene and how does that work? I’m then still going back to vitamin D and asking do we know everything about what it is converted to? And the answer is ‘no, we don’t.’ ”
Pike says it’s been very rewarding to pursue his vitamin D research at UW-Madison and be part of a legacy started by Steenbock and continued by DeLuca. While he continues his work on the vitamin’s role in gene regulation, he adds DeLuca himself is still marching ahead with new discoveries as well.
“Hector has outlasted everyone and he’ll probably outlast me,” he says. “He’s strong and going strong and because of his many basic as well as therapeutic advancements, he’s made himself and this department a real exemplar in this field.”

